


JOHN LI, M.D.
OTOLOGY NEUROTOLOGY RESOURCES
210 Jupiter Lakes Blvd #5105
Jupiter, FL 33458
Phone: (561)-748-4445
Fax: (561)-748-4449
Email: Dr-Li@Dr-Li.net
Esteem: Overview Esteem: FAQ Esteem: Fact Sheet How It Works / Testimonials
Envoy Esteem® Implant
Envoy Medical has produced the world's first FDA approved totally implantable hearing device. It is called the “Esteem.” The Esteem® is a breakthrough in technology that actually utilizes your own eardrum as the sound collection mechanism rather than using a synthetic microphone. The results are truly amazing in terms of sound clarity, quality and ease of use.
The device is completely implanted and completely invisible. No one will notice that you have an Esteem® implanted. There is no need to wear your hair long to cover your ears.


Ease-of-use
The Esteem® requires no maintenance. There is no device to take out and put on the nightstand. There are no worries about getting any electronics wet. You can swim, shower and exercise without worry. The batteries do not need to be recharged every night. It simply keeps working and working for years on end. The batteries are tested to last approximately 6 or 7 years if used 16 hours a day for seven days a week. (Continuous use at very high noise levels may reduce battery life. The range is 3-9 years) At the end of that time, a minor procedure is necessary to replace the battery.
Sound Quality
The sound quality and level of amplification has been reported by patients to be amazing. Because it uses the human eardrum as a microphone, the sound collection is more natural. Thus, the sound experience is much more natural. There is no "squealing" feedback that plagues conventional hearing aids usage. Most patients who have previously tried hearing aids are thrilled with the improvement from the Esteem.
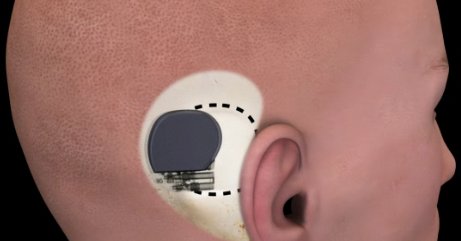
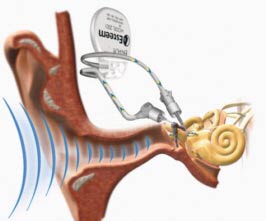
Surgery
The surgery is a very technically complex procedure that involves an incision behind the ear, and a mastoidectomy approach. A pocket is made for the battery/receiver unit. Next the hearing bones are prepared to accept the implants. A section of the incus bone is removed with a laser in order to redirect the incoming sound to the receiver, so that it can be amplified and be converted to a more powerful vibration that is sent to the stapes bone via the “driver” device. The "sensing” device and the "driver" device are carefully positioned within the mastoid cavity so that the optimal vibration sensitivity is achieved. The devices are then cemented into place, and then the system is tested with specially calibrated lasers that predict an excellent outcome. (If not, adjustments are generally made until an excellent outcome is achieved.) There are cases in which the implant is surgically found to be incompatible. In those rare cases, the procedure is terminated before any major changes are made in the hearing bones connections.
The postoperative period is usually uneventful although some patients have some discomfort just as they would have with any other surgery. Patients who have flown into town (usually for the day before surgery) are usually allowed to leave two or three days after surgery.
Activation
Activation is performed by the Envoy field clinical engineer two months after surgery. Some tests are run to determine device integrity. Once that is done, the device is activated, generally at an initially lower level. Adjustments may be made later on to bring the levels higher. That avoids the sudden discomfort that one might experience when something is suddenly too loud. That would be analogous to the visual discomfort noted when coming out of the dark room into a brightly lit room.
Esteem: Overview Esteem: FAQ Esteem: Fact Sheet How It Works / Testimonials